Synthetic polymers
Many organic molecules can be made to join up with one another to form very long molecules which can be thousands of atoms long. This makes new materials with water resistant properties. Many of these materials will soften when heated and can therefore be easily moulded into complex shape.These materials are known are therefore "thermoplastic" - often abbreviated to "plastic".
Students should:
- 4.44 know that an addition polymer is formed by joining up many small molecules called monomers
Addition polymers are made ( synthesised) by taking a large number of small molecules and joining them together to make long chain molecules.
The small molecules are known as monomers. The long chains are known as polymers.
Students should:
- 4.45 understand how to draw the repeat unit of an addition polymer, including poly(ethene), poly(propene), poly(chloroethene) and (poly)tetrafluoroethene
- 4.46 understand how to deduce the structure of a monomer from the repeat unit of an addition polymer and vice versa
Watch the video carefully and notice how the monomers can be turned into polymers.
Enter your text here ...
Enter your text here ...
Students should:
- 4.47 explain problems in the disposal of addition polymers, including: • their inertness and inability to biodegrade • the production of toxic gases when they are burned
- plastic pollution. non bio degradability leads to serious pollution
- many are sensitive to ultra violet radiation and degrade in sunlight
- can be flammable and toxic when burned
- mostly made from fossil fuels
- water resistant, can be used for bottles and buckets
- poor conductors of heat and electricity - used as insulators
- cheap and cost effective
- many are thermoplastic and can be moulded easily
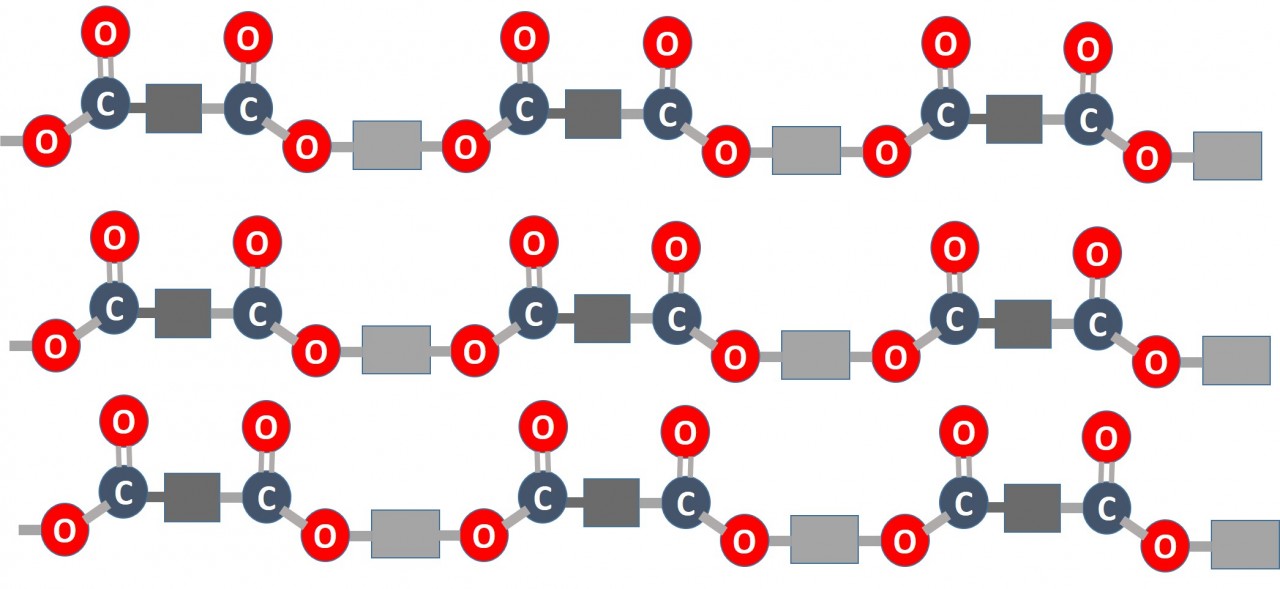
Condensation Polymerisation
Enter your custom HTML codes in this section ...
| Name of Monomer | Name of polymer | Common name of polymer | Properties and uses |
| ethene |
polyethene | polythene(HDPE)&(LDPE) | the most common type of plastic |
| chloroethene | polychloroethene | PVC glue | water pipes and electrical cable insulation |
| propene | polypropene | polypropylene | crates and ropes |
| phenylethene | polyphenylethene | polystyrene | packing, insulation, plastic cups |